Malte Drescher / Marcel Leist / Christine Peter
Local and transient structural features of the intrinsically disordered protein alpha-synuclein
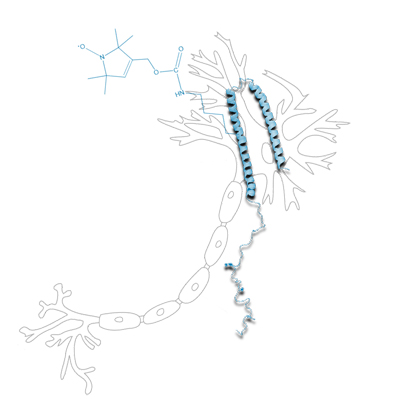
The classical concept of proteostasis assumes a steady state between folding and misfolding of proteins. This picture gets considerably more complex for intrinsically disordered proteins (IDPs), since they exist in multiple conformational states. The Parkinson protein α-synuclein (ASYN) is a model system among the IDPs. A traditional view is that ASYN is disordered in solution and can adopt a variety of stable conformations upon macromolecular interaction. However, findings that ASYN assumes structured features already in solution, suggest an alternative hypothesis for its role in proteostasis that will be investigated in the present project by combining molecular simulation, spectroscopy, and biological in vitro experiments.